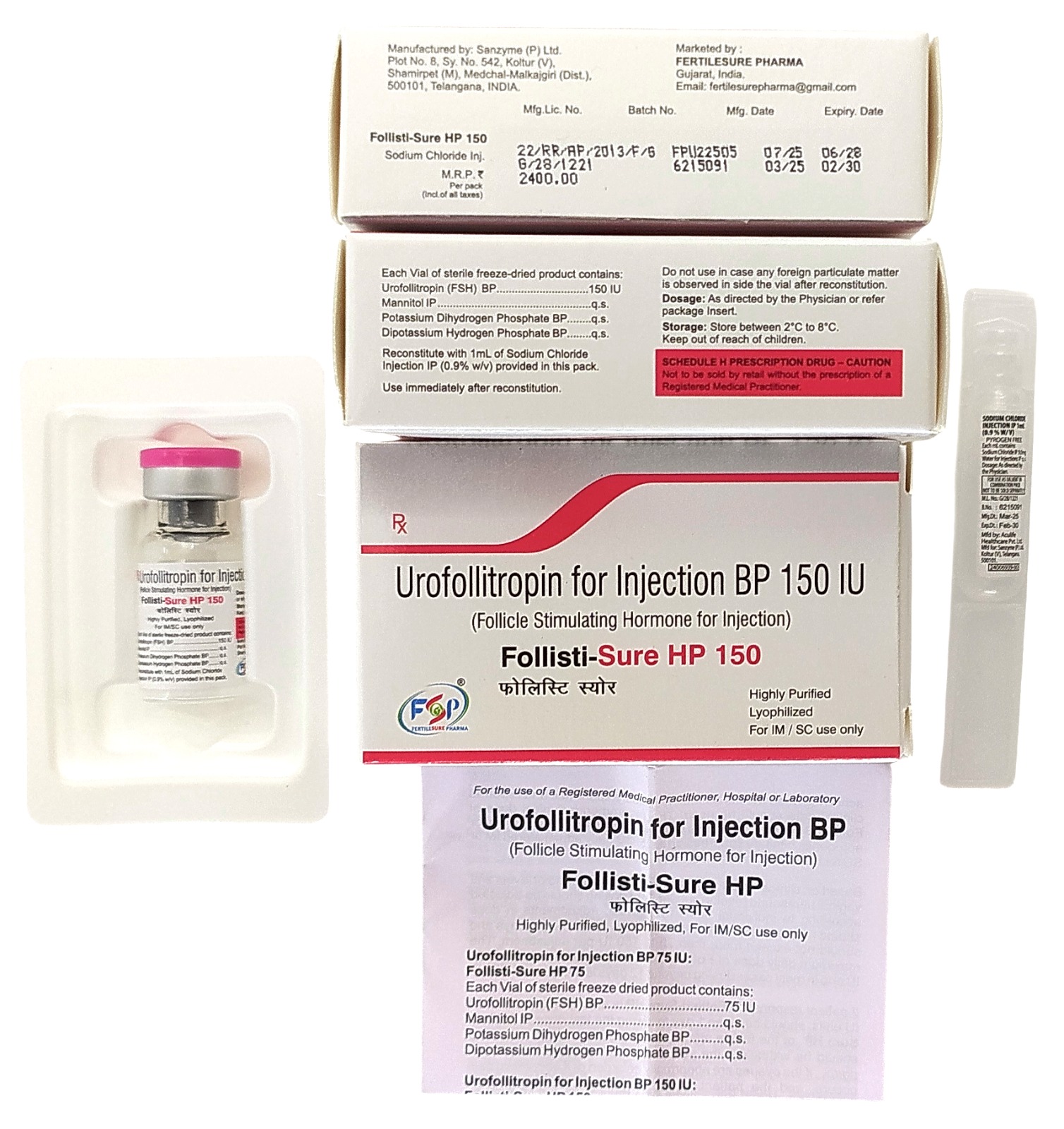
Urofollitropin for Injection BP 150 IU
$300.00
SKU : N/A
Category : Hormonal
Urofollitropin for Injection BP 150 IU is a prescription medicine containing human follicle-stimulating hormone (FSH), used under medical supervision in reproductive function support in infertility treatments.
Urofollitropin for Injection BP 150 IU
Product Overview
Urofollitropin for Injection BP 150 IU is a prescription hormonal medicine used in the management of certain infertility conditions. It contains urofollitropin, a form of human follicle-stimulating hormone (FSH), which is involved in reproductive function. This medicine is prescribed as part of assisted reproductive treatment protocols under medical supervision.
How It Works
Urofollitropin functions similarly to follicle-stimulating hormone in the body. In females, FSH is involved in the growth and maturation of ovarian follicles. In males, it acts on the testes to support sperm production. These actions are related to reproductive processes when natural hormone levels are insufficient.
Uses / Indications
Urofollitropin for Injection BP 150 IU is indicated for:
- Treatment of female infertility associated with ovulatory dysfunction
- Controlled ovarian stimulation in assisted reproductive techniques such as in vitro fertilization (IVF)
- Treatment of male infertility related to hypogonadotropic hypogonadism as part of hormone therapy
Use is based on individual patient evaluation and is not intended for self-medication or non-medical use.
Composition
- Active Ingredient: Urofollitropin (Human Follicle-Stimulating Hormone)
- Dosage Form: Powder for solution for injection
- Strength: 150 IU per vial
The formulation is intended for reconstitution prior to administration and is manufactured according to pharmacopeial standards (BP).
Administration
Urofollitropin for Injection is administered via subcutaneous or intramuscular injection as directed by a healthcare professional. Treatment protocols, duration, and monitoring depend on the patient’s condition and response to therapy. Clinical monitoring, including hormonal assessments and imaging where appropriate, may be required.
Storage Instructions
- Store below 25°C
- Protect from light
- Do not freeze
- Keep out of reach of children
- Use only if the solution is clear and free from visible particles after reconstitution
- Follow the storage instructions provided on the product packaging
Important Information
- Use only under medical supervision
- Improper use may increase the risk of adverse effects, including ovarian hyperstimulation in women
- Not suitable for patients with hormone-dependent tumors, unexplained vaginal bleeding, or primary ovarian failure
- Medical evaluation and monitoring are required during treatment
For general information on fertility hormones and their medical use, refer to:
https://medlineplus.gov/infertility.html
| Pack Size |
6 Injection |
|---|
Related products
Calcium Citrate Malate, Vitamin D3 & Zinc Chewable Tablets
Calcium Citrate Malate, Vitamin D3 & Zinc Chewable Tablets are used as a daily supplement for bone health and nutritional support.
Chorionic Gonadotrophin Injection 5000 IU1mL
Chorionic Gonadotrophin Injection 5000 IU/1mL contains hCG and is used under medical supervision in fertility treatment, ovulation induction, and reproductive hormone management.
Chorionic Gonadotrophin Injection IP 10000 IU
Chorionic Gonadotrophin Injection IP 10000 IU contains chorionic gonadotrophin and is used under medical supervision in ovulation induction and hormone management.
FERTIPARIN 40
FERTIPARIN 40 is used under medical supervision in assisted reproductive treatments and blood flow management.
FERTIPARIN 60
FERTIPARIN 60 contains low molecular weight heparin and is used under medical supervision in the prevention and management of blood clot-related conditions.
L-Arginine & Proanthocyanidin Granules Sachets
L-Arginine & Proanthocyanidin Granules Sachets is used under medical supervision in circulation and vascular function support, containing an amino acid and antioxidants.
Menotropin For Injection IP 1200 IU
Menotropin For Injection IP 1200 IU contains human menopausal gonadotropins (FSH and LH) and is used in fertility and hormonal treatments under medical supervision.
supervision.Urofollitropin for Injection BP 75 IU
Urofollitropin for Injection BP 75 IU contains follicle-stimulating hormone (FSH) and is used under medical supervision in ovarian follicle development during fertility treatments.