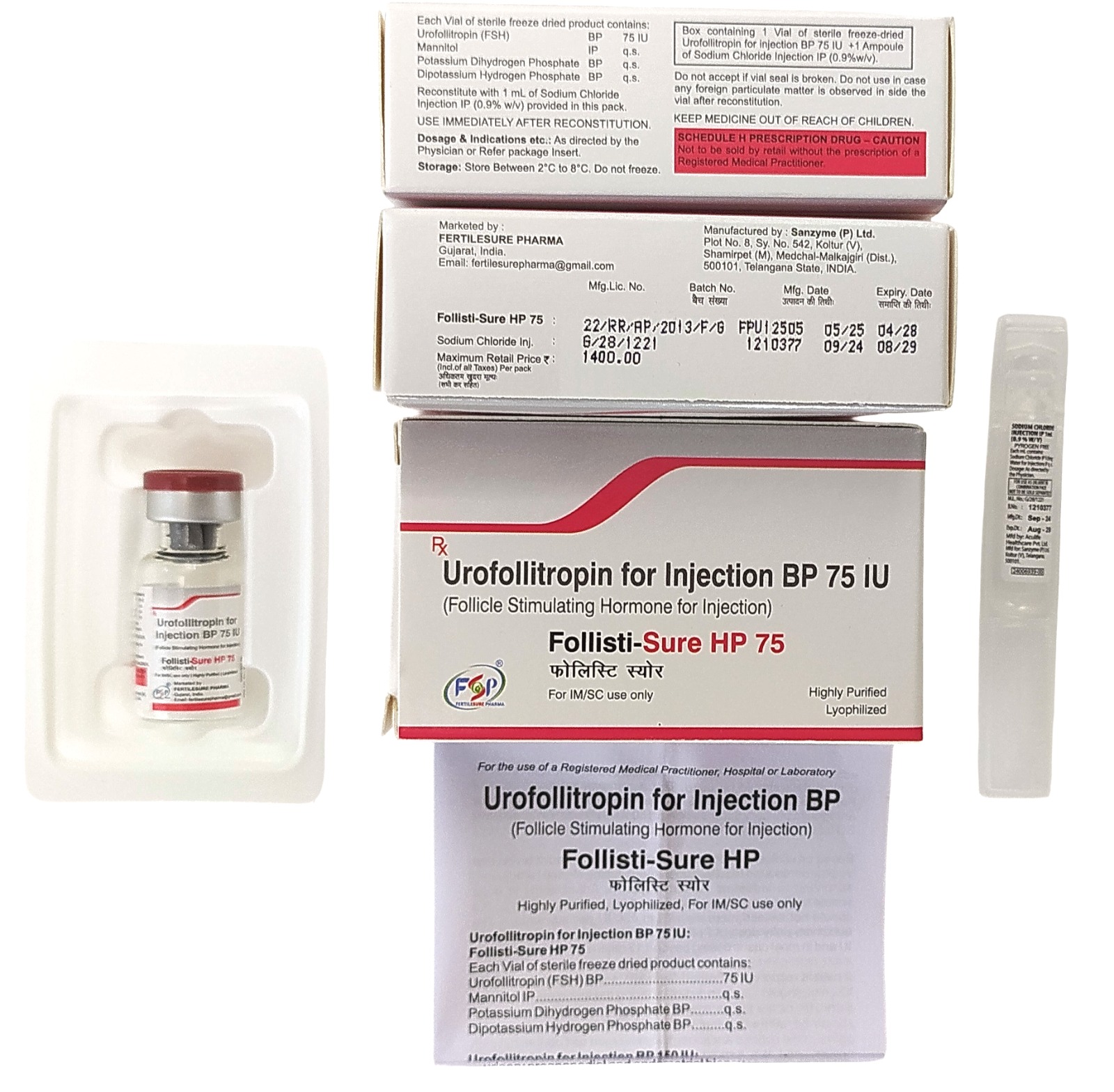
Product Overview of Urofollitropin for Injection BP 75 IU
Urofollitropin for Injection BP 75 IU is a prescription medication containing a purified form of follicle-stimulating hormone (FSH). It is primarily used in assisted reproductive therapies to support ovarian follicle development in women undergoing fertility treatments. Administered under strict medical supervision, it helps stimulate the production of mature eggs for procedures such as in vitro fertilization (IVF) or intrauterine insemination (IUI).
How Urofollitropin for Injection BP 75 IU Works
The active ingredient, urofollitropin, mimics the body’s natural FSH. FSH regulates the growth and maturation of ovarian follicles. When it is administered, it binds to ovarian cell receptors, stimulating follicular development and estrogen production, which is essential for ovulation. This action supports natural reproductive processes and increases the likelihood of successful egg retrieval during fertility treatments.
Uses / Indications of Urofollitropin for Injection BP 75 IU
This is indicated for women with fertility challenges due to low or absent FSH activity, including certain types of anovulation or hypogonadotropic hypogonadism. It is also used in controlled ovarian hyperstimulation protocols for assisted reproductive technologies such as IVF or IUI. Treatment should always be initiated and monitored by a qualified healthcare professional.
Composition of Urofollitropin for Injection BP 75 IU
Each vial of Urofollitropin for Injection contains 75 IU of urofollitropin as the active ingredient. The medicine comes in a sterile, injectable powder form that must be reconstituted with the supplied solvent prior to use. The formulation meets British Pharmacopoeia (BP) standards, ensuring purity, quality, and consistency.
Administration of Urofollitropin for Injection BP 75 IU
This is administered via subcutaneous or intramuscular injection. The dosage and treatment duration are tailored to each patient based on clinical evaluation and response. Regular monitoring of hormone levels and ovarian follicle growth through ultrasound is essential to ensure safety. Self-administration should only be performed after receiving proper training from a healthcare professional.
Storage Instructions for Urofollitropin for Injection BP 75 IU
Store Urofollitropin for Injection BP 75 IU in a refrigerator at 2°C to 8°C (36°F to 46°F). Do not freeze. Protect from light and moisture. After reconstitution, use the solution immediately or follow storage instructions provided by your healthcare provider. Keep all medications out of reach of children.
Important Information / Warnings for Urofollitropin for Injection BP 75 IU
Use Urofollitropin for Injection BP 75 IU strictly under medical supervision. Possible risks include ovarian hyperstimulation syndrome (OHSS), multiple pregnancies, and injection site reactions. Patients should report any unusual symptoms such as abdominal pain, rapid weight gain, or severe bloating to their doctor immediately.
This medication is not suitable for women with ovarian cysts unrelated to fertility treatment, unexplained vaginal bleeding, or known hypersensitivity to gonadotropins. For more information on safe fertility treatments, visit MedlinePlus – Fertility Medications.
| Pack Size |
6 Injection |
|---|
Related products
Carboprost Tromethamine Injection I.P.
Chorionic Gonadotrophin Injection IP 10000 IU
FERTIPARIN 60
L-Arginine & Proanthocyanidin Granules Sachets
Leuprolide Acetate for Injection (Depot 3.75 mg)
Leuprolide Acetate for Injection is a long-acting GnRH agonist used to manage hormone-dependent conditions by suppressing estrogen or testosterone levels under medical supervision.